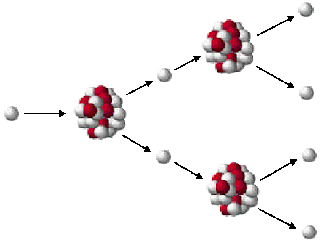
Einstein’s equation connects two seemingly distinct things – mass which is solid and energy in the form of heat. Deuterium and tritium (isotopes of hydrogen) can be fused to produce helium and at the same time, the lost mass converts into energy. The leftover mass becomes energy in the form of heat. The process releases energy because the total mass of the resulting single nucleus is less than the mass of the two original nuclei. In a fusion reaction, two light nuclei merge to form a single heavier nucleus. As in fission, the fusion process also produces a massive amount of heat as the weight of the new atom is less than the weight of the fused atoms but also produces much less radioactivity. The opposite process to fission is ‘fusion’ in which two atoms are pushed together through the use of heat and pressure to create a new and different atom. That can then be used to produce steam which turns a traditional turbogenerator set to produce power. In civil use, the energy released in the fission process is controlled within a pressure vessel and used to heat water or helium gas or sometimes liquid sodium. This 'missing' mass, only a tiny fraction of the original, has been converted into energy according to Einstein's equation. The ‘fission’ process which is the splitting a uranium atom into two fragments of slightly different atomic weight from the original fuel can release a huge amount of energy in the form of heat as the sum of the masses of these fragments is less than the original mass. 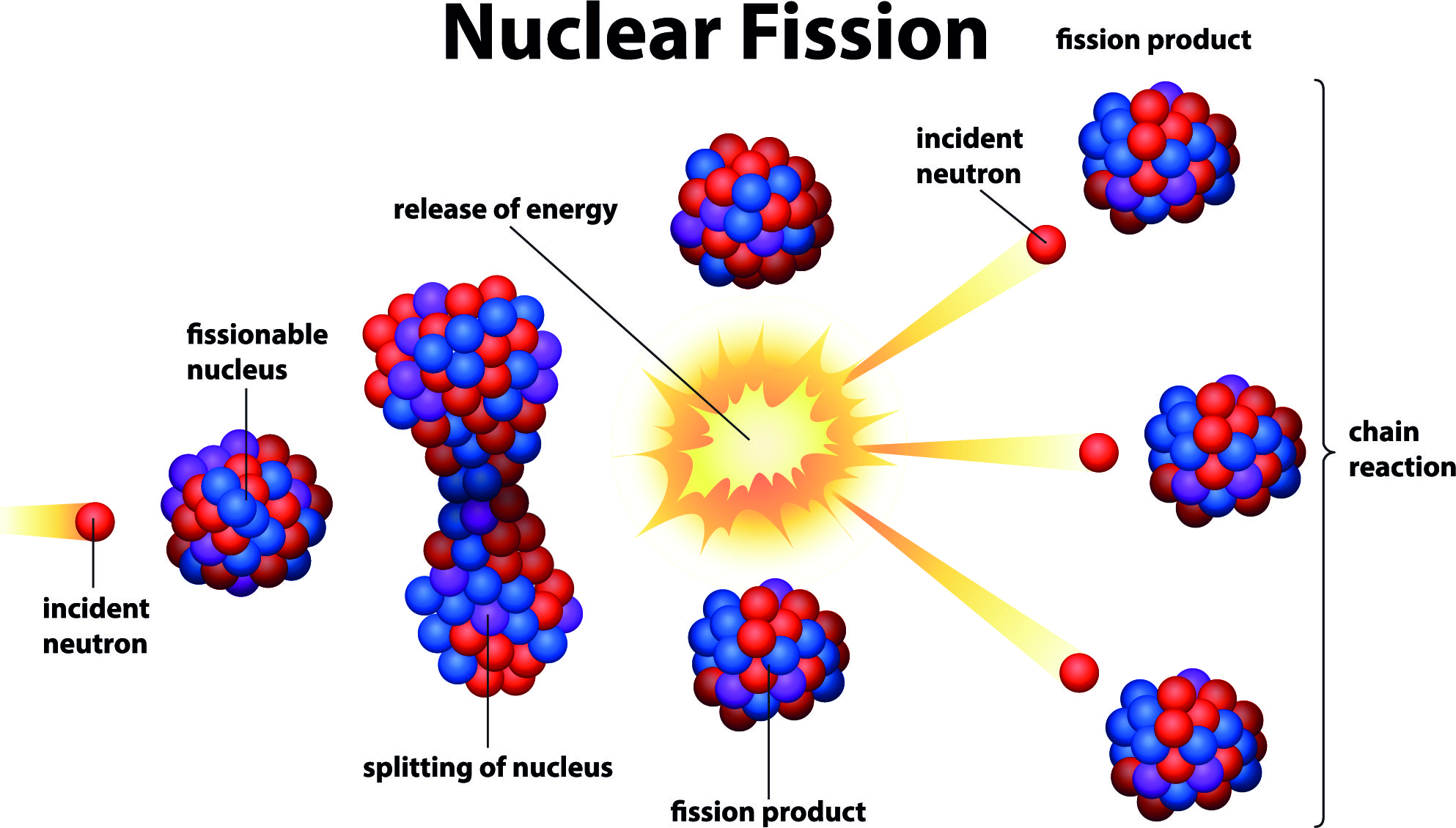
This equation explains the reason why the atomic bomb which contains only very small amounts of ‘fuel’ can produce so much energy. It puts the connection between energy and mass in mathematical language. Many of us will remember from our physics class at school, Einstein’s equation E = mc 2, sometimes called the world’s ‘most famous equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
